Q1. A 2.16 g of a substance occupies 1.5 cm3. Its density to appropriate significant figure is:
Q2. Find the option with 3 significant figures.
Q3. Significant figures depend on ________ number of significant figures in given data.
Q4. The method used to measure the distance of a planet from the Earth is
Q5. A dimensionless quantity
Q6. Magnitude of force F experienced by a certain object moving with speed v is given by F = Kv3, where K is a constant. The dimensions of K are
Q7. What is the dimension of (½)at2?
Q8. Relative error in Z, given that Z = 1/A2 is:
Q9. Parallax method is used to measure:
Q10. The order of magnitude of a number expressed in scientific notation is
Q11. The numbers of significant figures in 9.1 x10-31 kg are:
Q12. One angstrom is equal to __________ .
Q13. Approximate ratio of 1 fermi to 1 angstrom is ..............
Q14. The number of significant figures in the distance of one light year , 9.4605 × 1015 m is
Q15. Which of the quantity is dimensionless?
Q16. The most suitable instrument for measuring the size of an atom is.
Q17. Angstrom is the unit of
Q18. A vernier calipers is used for measuring lengths to an accuracy of:
Q19. Which unit system is presently internationally accepted?
Q20. A 5.2 g of a substance occupies 1.11 cm3. Its density to appropriate significant figure is:
Q21. The position of pen seems to be changed when you look at it through your left and right eye w.r.t to a fixed point on a wall. This effect is called ________ .
Q22. Find the option with 5 significant figures
Q23. Find the odd one out.
Q24. Three physical quantity having dimensions [M L-1 T-2] are
Q25. The numbers of significant figures in 1.84 x10-27 kg are:
Q26. Random errors occur due to:
Q27. Which of the following physical quantities has different dimensions?
Q28. The base units for length, mass and time in MKS system are:
Q29. 1 unified atomic mass unit is equal to.
Q30. Which of the following physical quantity is dimensionless?
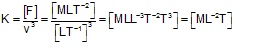
Comments
Post a Comment